|
|
|
Nematic Liquid crystals are rod like molecules that in the absence of external influences arrangeĀthemselves
parallel to each other and hence tend to point in a given direction. The average orientation of molecules is given by the director (n),
which is essentially a bi-directional vector which quantifies the orientation of the molecules in a given region of the sample. When nematic
liquid crystals are confined to a curved surface, the topological constraints imposed by the surface causes a distortion in the molecular
orientation. In certain regions, the molecular orientation is such that the director cannot be defined. Such regions are called defects.
|
|
We study topological defects that arise when nematic liquid crystals are confined to spherical geometries. One of
our motivations for studying these defects is that they are regions that can be functionalized and made to serve as "bonds" which
would allow the fabrication of self assembling particles with fascinating properties [1].
Experimental realization of these kinds of geometries is fascilitated by the use of microfluidic techniques with which
we fabricate double emulsions of nematic LC. A double emulsion is a drop inside a drop, and hence the resulting structure is a shell of LC.
By utilizing the control afforded by this technique in conjunction with osmosis, the size of the shells can be controlled to make them as
thin or as thick as desired. By making them extremely thin, we approach the limit where the shell can be considered a bi-dimensional surface.
Theory predicts that a bi-dimensional sphere of nematic LC should give rise to four defects of topological charge +1/2 located at the vertices
of a tetrahedron [2].
By studying ultrathin double emulsion shells, we have shown that four +1/2 defects are indeed located at the
vertices of a tetrahedron, as can be seen in Fig. 2.
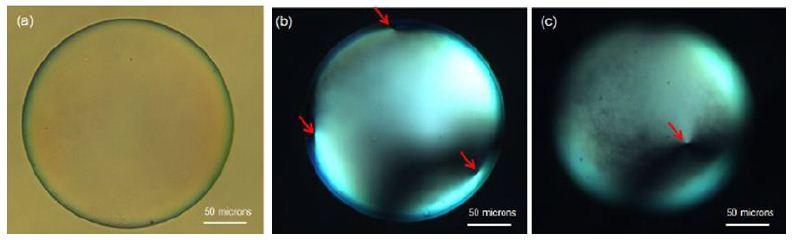
Fig. 2: (a) Brightfield image of an ultrathin shell (b) cross-polarizer image of top plane showing three defects (c)
cross-polarizer image of bottom plane showing four defects.
In addition to having four defects, ultrathin shells of the same size can have either three or two defects.
Three defect shells consist of two +1/2 defects and a +1 defect, and two defect shells consist of two +1 defects.
In ultrathin shells with three defects, we have shown that the defects are located at the vertices of an isosceles triangle,
as shown in Fig. 3. This is expected because topological defects in nematics interact via a long-range interaction which tends to
maximize the defect separations, resulting, in this case, in an isosceles structure because only two of the three charges are of the
same strength.

Fig. 3: (a) Image under cross-polarizers of an ultrathin shell
with three defects located at the vertices of an isosceles triangle.
In ultrathin nematic shells with two +1 defects, we have shown that the defects are located diametrically
opposite to each other, as shown in Fig. 4.

Fig. 4: (a) Image under cross-polarizers of an ultrathin shell
with two defects located diametrically opposite each other.
We are currently looking at ways in which the dimensions of the shells influence the formation of either the four,
the three or the two defect structure.
References:
- [1] D. R. Nelson, Nano Lett. 2, 1125 (2002).
- [2] T. C. Lubensky and J. Prost., J. Phys. II (France) 2, 371 (1992).
- [3] D. R. Nelson, Defects and Geometry in Condensed Matter Physics (Cambridge University Press)
- [4] G. A. DeVries et al., Science 315, 358 (2007).
- [5] V. Vitelli and D. R. Nelson, Phys. Rev. E 74, 021711(2006).
- [6] A. Fernandez-Nieves, V. Vitelli, A. S. Utada, D. R. Link,
M.Marquez, D. R. Nelson, and D. A. Weitz, Phys. Rev. Lett. 99, 157801 (2007)
- [7] T. Lopez-Leon and A. Fernandez-Nieves, PhysRevE.79.021707 (2009)
- [8] G. Skacej and C. Zannoni, Phys. Rev. Lett. 100, 197802 (2008)
- [9] H. Shin, M. J. Bowick, and X. Xing, Phys. Rev. Lett. 101,037802 (2008)
- [10] M. A. Bates, J. Chem. Phys. 128, 104707 (2008)
- [11] A.S. Utada, et.al. Science 308, 537 (2006)
Contact Information:
Sharan Devaiah
Office: Boggs Building, Room B-54
Email address: sharan [at] gatech.edu
Phone: 404-385-3681
|
|